- [email protected]
- +44 (0) 23 8048 3000
Calprotectin and Clinical Correlation
- Resource: fCAL turbo
- Date: February 21, 2018
Mary O’Connell née Deasy, Biochemistry Department, Mercy University Hospital, Cork
 In issue 2017-3 of Leading Edge, Mary O’Connell detailed her evaluation of the BÜHLMANN fCAL® turbo assay on the Abbott Architect C8000 analyser in comparison to the Phadia 250 ELiA™ Fluorenzyme-Immunoassay.
In issue 2017-3 of Leading Edge, Mary O’Connell detailed her evaluation of the BÜHLMANN fCAL® turbo assay on the Abbott Architect C8000 analyser in comparison to the Phadia 250 ELiA™ Fluorenzyme-Immunoassay.
Data was collected from 60 patient faecal samples that were tested for calprotectin concentrations using both methods.
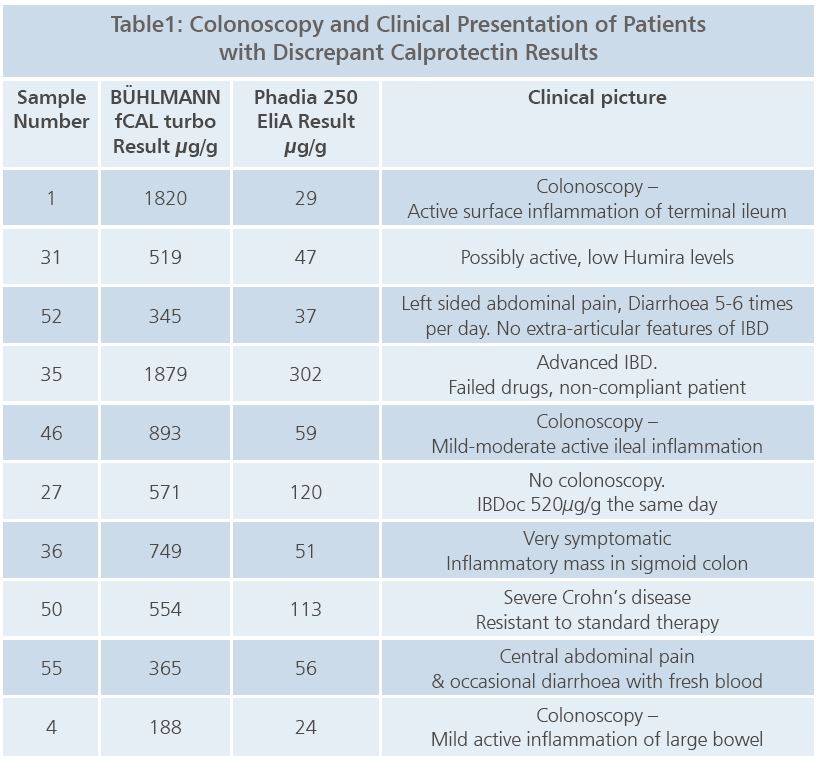
All 60 results from the two methods were reviewed by the IBD medical team. Twenty two of the samples produced discrepant results between the two testing methods. The patient charts of these 22 discrepant results were reviewed for colonoscopy reports, if they were performed, and clinical presentation, to help determine which of the assays produced the more accurate results.
Table 1 shows examples of the main patient samples that were used to make the decision that the BÜHLMANN fCAL turbo correlated better to the clinical picture. In these cases the higher calprotectin concentrations measured with fCAL turbo were associated with a corresponding medical status.
The medical team concluded that the fCAL turbo results correlated very well with the participant’s clinical presentation and stage of IBD. This is important as the IBD team use this data along with the clinical presentation before referring patients for colonoscopy.