- [email protected]
- +44 (0) 23 8048 3000
Out of Africa: Using Faecal Calprotectin Assays in the African Setting and Screening for Intestinal Schistosomiasis
- Resource: Quantum Blue
- Date: March 12, 2018
 Professor Russell Stothard and Dr Amaya Bustinduy
Professor Russell Stothard and Dr Amaya Bustinduy
Liverpool School of Tropical Medicine, London School of Hygiene and Tropical Medicine
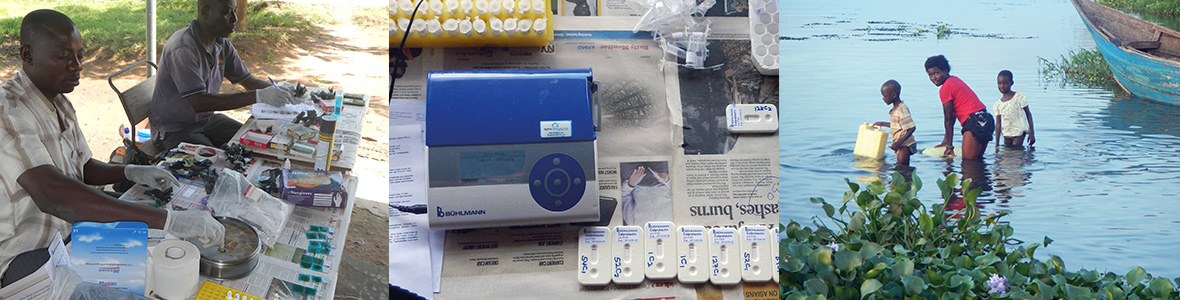
Our article highlights ongoing work in Uganda measuring morbidity associated with intestinal schistosomiasis using faecal calprotectin tests. Our original study was as first reported in PLOS NTDs see http://journals.plos.org/plosntds/article?id=10.1371/journal.pntd.0002542
Expanded access to point-of-care tests (POC) is important for screening a variety of patient disorders, for example, it allows earlier detection of conditions that might benefit from prompter clinical intervention(s). From a public health perspective the test might also be useful to monitor the general health of a targeted demographic group such as school children.
Here in the UK, the BÜHLMANN Quantum Blue® lateral flow faecal calprotectin test has been useful in the management of common intestinal aliments such as inflammatory bowel disease (IBD) and has also been used in local GP-level settings. This POC assay measures concentrations of faecal calprotectin in the stool which is typically resultant from activated neutrophils that have been recruited into the gastrointestinal surface, indicative of a diseased state.
The technology behind this assay is portable, robust and simple to perform without recourse to complicated preparation or liquid handling steps. As such these factors also make the assay useful for re-purposing in disease surveillance in other parts of the world, especially where inappropriate fieldbased technology induces a health screening bottleneck.
In sub-Saharan Africa (SSA), for example, very little is known about the general levels of faecal calprotectin within the healthy populous, nor how raised levels may signpost gastrointestinal disease. Notably in many parts of SSA, intestinal schistosomiasis maybe be very common, infecting over 200 million people, and leads to a variety of gastrointestinal disorders.
These include active lesions on the mucosal surface that bleed, giving rise to trace amounts of blood in the stool.
To read the full article, click here