- [email protected]
- +44 (0) 23 8048 3000
Case Studies / fCAL turbo
Liquid Elastase Samples – To Test, or Not to Test? That is The Question…
Alison Jones, Consultant Clinical Biochemist, York and Scarborough NHS Foundation Trust It is well documented that liquid/watery stool samples should not be tested for faecal elastase due to the risk
Switching things up… Transitioning the BÜHLMANN fCAL® turbo for Calprotectin Testing
From the Siemens Advia 2400 to the Atellica Analyser Georgette Glover, Senior Biomedical Scientist, Biochemistry Department, Addenbrookes Hospital Addenbrookes Hospital has been using the BÜHLMANN calprotectin assays for a number
Building on the Success of fCAL turbo… CALEX® Cap Brings Improved Workflow and New Multi Assay Functionality
As you may have seen from the NEQAS reports, the BÜHLMANN fCAL® turbo calprotectin assay is proving very popular, and its use has grown significantly over the last few years.
Switching from Phadia to BÜHLMANN for a More Efficient Calprotectin Service
The Newcastle upon Tyne Hospitals Foundation Trust is one of the largest NHS trusts in the UK, covering six hospitals. Elodie and her team in the department of blood sciences
Using the BÜHLMANN fCAL® turbo on Beckman Coulter AU5800 – A Report from SYNLAB
A Report of A. Suter* and Dr. C. Fuhrer** from SYNLAB *BMA HF/ Fachleiterin, **Head of Core Lab, FAMH Clinical Chemistry, NF hematology, microbiology and immunology; SYNLAB Suisse SA, Alpenquai
Experience of the BÜHLMANN fCAL® turbo on DxC 800, Beckman Coulter
A Report of Martina Kobelt* and Karin Jung**, Zentrum für Labormedizin, KS St. Gallen, Switzerland *Senior Laboratory Technician Core Lab, **Deputy Head of Department Core Lab/FAMH Candidate Clinical Chemistry, Centre
Unilabs Portugal using BÜHLMANN fCAL® turbo in routine
Unilabs Portugal, a private European lab chain, made a comprehensive evaluation of different Calprotectin assays on the market. Finally, they choose the BÜHLMANN fCAL® turbo simply because it is faster,
From BÜHLMANN Quantum Blue® to fCAL turbo®
Follow the Evolution of Calprotectin Testing at James Cook University Hospital This interview with Mr Andrew Teggert, Clinical Scientist, James Cook University Hospital, Middlesbrough, discusses recent changes made to the faecal calprotectin testing
Calprotectin and Clinical Correlation
Mary O’Connell née Deasy, Biochemistry Department, Mercy University Hospital, Cork In issue 2017-3 of Leading Edge, Mary O’Connell detailed her evaluation of the BÜHLMANN fCAL® turbo assay on the Abbott Architect C8000 analyser in comparison
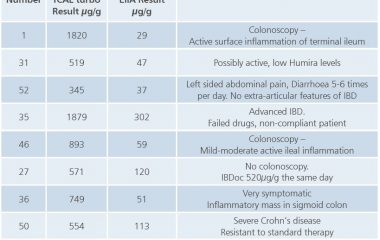
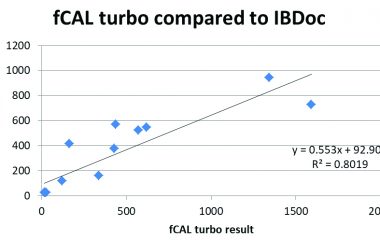
Calprotectin Home vs. Laboratory Test
Mary O’Connell née Deasy, Biochemistry Department, Mercy University Hospital, Cork, Ireland The Mercy University hospital is one of the largest users of the IBDoc test, with almost 500 patients using the system to